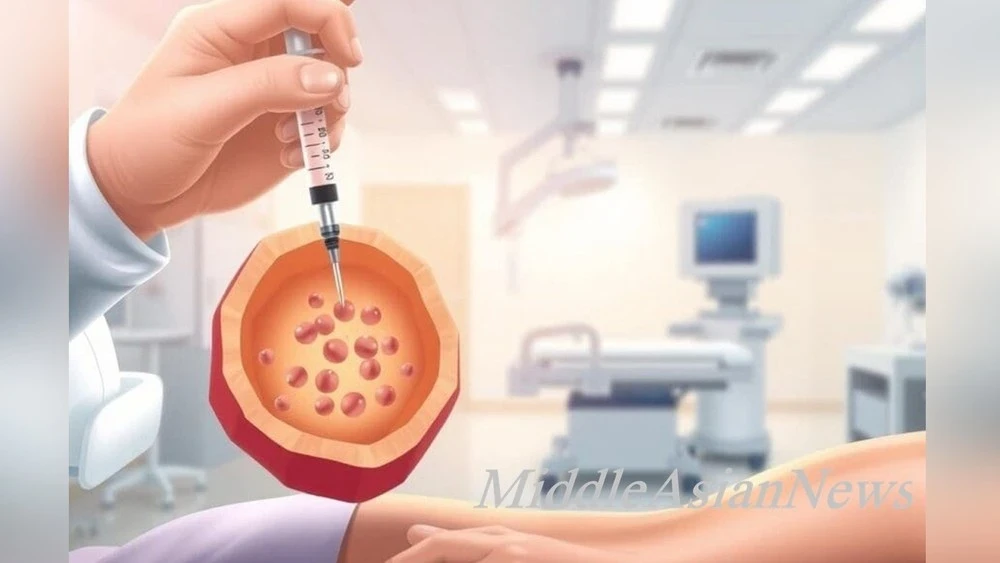
The Japanese Ministry of Health has given the green light for the commercial use of revolutionary regenerative medicine drugs based on induced pluripotent stem cells (iPS). These developments are intended for the therapy of serious diseases such as Parkinson's disease and heart failure, reports Kazinform, citing SCMP.
In particular, this concerns the ReHeart drug, used for the treatment of ischemic cardiomyopathy, and Amchepry, which is used to correct degenerative changes in Parkinson's disease.
Shinya Yamanaka, Nobel laureate and honorary director of the Kyoto University iPS Cell Center, expressed his joy at this event: "I am very pleased to see the first major step towards the practical implementation of the technology 20 years after its discovery."
The ReHeart method involves the use of cardiomyocytes derived from iPS cells, which are applied to the patient's heart. This helps stimulate angiogenesis and restore organ function. Clinical trials have shown that patients experience a reduction in symptoms of shortness of breath and an improvement in physical endurance.
As for Amchepry, it involves the introduction of precursor cells into the brain that begin to produce dopamine. During the trials, six out of seven patients showed partial restoration of motor functions.
However, the approval is limited and will be valid for seven years. The drugs can only be used in small clinical groups. Full implementation is possible only after confirming their safety and efficacy within the specified timeframe.
It is expected that sales of these drugs may start by the end of the year, when pricing procedures and inclusion in the health insurance system are completed. The company Cuorips has already announced its plans to begin sales this year.